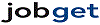Average salary: $84,238 /yearly
More statsSearch Results: 1,370 vacancies
US Regulatory Affairs Lead
-------------------------------------------------------------------------------------------------
SCOPE OF RESPONSIBILITIES:
" He/She will be responsible for representing regulatory function to approve promotional material...
Regulatory
...Regulatory Affairs Specialist
The Regulatory Affairs Specialist IV will coordinate the accumulation of technical information and prepare submissions for in vitro diagnostic and medical device licenses to various governmental regulatory agencies. They will also coordinate...
Regulatory
...CorDx, a thriving biotechnology company, is seeking a Regulatory Coordinator to join our team. In this role, you will assist in the planning and execution of regulatory compliance activities. You will analyze and interpret regulations and guidelines to ensure company...
Regulatory
...experience, and who share a common goal of getting our technology into every physician’s office. We have a great opportunity for a Regulatory Affairs Specialist to join our team!
ABOUT THE ROLE
The Regulatory Affairs Specialist will assist in regulatory submissions,...
Regulatory
Flexible hours
...[Full Time] Cybersecurity Regulatory Specialist $125k at MedCrypt (United States) | BEAMSTART Jobs Cybersecurity Regulatory Specialist $125k
MedCrypt United States
Date Posted
06 Apr, 2023
Work Location
San Diego, CA, United States
Salary Offered...
Regulatory
Full time
Contract work
Part time
Internship
Remote job
Flexible hours
...protected status.
Description: Position Summary
This position is responsible for the development and implementation of regulatory strategies and FDA regulated quality assurance activities for pipeline programs to ensure compliance with applicable regulations...
Regulatory
Full time
Afternoon shift
...Job Description
Job Description Regulatory Affairs Specialist
Monday – Friday, 8:30 am – 5:00 pm
About Us
At Argen, we’re in the business of creating healthy, confident smiles one case at a time. Argen is a global leader in dental manufacturing with headquarters...
Regulatory
Holiday work
For contractors
For subcontractor
Local area
Worldwide
Monday to Friday
...San Diego, CA. Acadia's hybrid model requires this role to work in our office three days per week on average.
The Manager of Regulatory Affairs (RA) Labeling will assist in the development of a cross-functional labeling strategy for products worldwide, including providing...
Regulatory
Holiday work
Local area
Remote job
Worldwide
Flexible hours
Night shift
3 days per week
...Job Title
Regulatory Affairs Specialist
Job Description
Looking at the challenges the world is facing today Philips' purpose has never been more relevant. So, whatever your role, if you share our passion for helping others, you'll be working towards creating...
Regulatory
Worldwide
Work visa
...Job Description
Job Description Description: Responsible for providing support in Regulatory Operations filing multiple original INDs each year and managing multiple active INDs in Phase 1/2 clinical trials (US, Canada, EU and APAC). He/she will manage day to day...
Regulatory
...goal of delivering diagnostic solutions to some of the most critical questions in healthcare.
Job Type: Full time Job Title: Regulatory Specialist - IVD and 510(k)
Location: Onsite - San Diego Office
Salary range: $ 60,000 to $100,000
Department: Regulatory...
Regulatory
Full time
...to improving patients’ lives by advancing novel therapies to address unmet medical needs.
Position Overview:
As the Head of Regulatory and Quality, you will be a key member of our executive leadership team, responsible for providing strategic direction and...
Regulatory
Full time
Relocation
...innovators who are collaborating to bring new therapies to patients in need.
The Position
Arrowhead is seeking an experienced Regulatory/Medical Writer to lead its Medical Writing Team. The Regulatory/Medical Writer will work collaboratively with Arrowhead's...
Regulatory
For contractors
...The role involves providing global regulatory oversight and project leadership for assigned development products, reporting directly to the Vice President of Regulatory Affairs. The incumbent will contribute to and execute regulatory strategies to facilitate global product...
Regulatory
**Job Description Summary**
As Staff Regulatory Affairs Specialist, Infusion, you will be responsible for Regulatory Affairs projects within the Medication Management Systems Infusion portfolio. Regulatory Affairs activities, will include: development, execution and...
Regulatory
Remote job
Work alone
Shift work
$99.75k - $123.9k
Extended Deadline: Tue 5/14/2024
UC San Diego values equity, diversity, and inclusion. If you are interested in being part of our team, possess the needed licensure and certifications, and feel that you have most of the qualifications and/or transferable skills for...
Regulatory
Hourly pay
Contract work
Local area
Remote job
$135k - $165k
...Commission Accredited institution with Disease-Specific Certification in CKD, VAD, and Comprehensive Stroke, is seeking an Ambulatory Regulatory Compliance Specialist to join our dynamic team. Recognized among the top 10 in quality and safety among academic medical centers,...
Regulatory
Hourly pay
Local area
...projects move through the environmental compliance process? Do you have a thorough knowledge of the southern California ecological regulatory environment?
Great Ecology is looking for a natural resource-focused Regulatory & Permitting Specialist to join an...
Regulatory
Holiday work
Relocation
...turnover, train new staff, oversee knowledge transfer between staff and prepare manuals of operations for applicable projects.
2) Regulatory compliance
The postdoc will carry out experiments in compliance with institutional, local, national and international...
Regulatory
Local area
Senior Regulatory Consultant
Part-Time
Cello Therapeutics, Inc. is a rapidly growing biotech company located in the Sorrento Valley—one of the major biotech hubs in the nation. Cello focuses on the development of novel nanotherapeutics based on biomimetic cell membrane...
Regulatory
Part time